
[ad_1]
I’ve beforehand regarded on the pigments used to color the E-book of Kells, which dates from round 800 AD and which contained arsenic sulfide because the yellow colourant. The Bayeaux tapestry is a later embroidery courting most likely from round 1077 and right here the colors are primarily based fully on mordanted pure dyes. These are usually acknowledged to be blue woad (precept element indigo), pink madder (precept element alizarin) and the much less well-known yellow weld, which comes from the plant Reseda Luteola and the precept element of which is luteolin.
Luteolin has an fascinating chemical historical past. It was first purified in 1829, within the daybreak of natural chemistry, and its components C15H10O6 established by 1864. A. G. Perkin, the son of the William Perkin who found the dye mauveine, then supplied the chemical construction[1] in 1896. This latter article is properly value a contemporary learn, because it fantastically illustrates how the artwork of construction dedication was performed within the days earlier than crystallography and NMR.
Perkin obtains his construction by evaluating luteolin to then recognized quercetin, concluding that the previous should additionally include an fragrant hydroxy group “ortho” to the carbonyl group, as in querecetin. The key experimental proof was that alkylation of luteolin with iodoethane solely produces a triethoxy by-product of luteolin, with “one hydroxy group resisting ethylation“. It was by then established, by 4 completely different units of researchers, that hydroxy teams adjoining to the carbonyl in e.g. quercetin or alizarin resisted alkylation. The construction of luteolin was established (see eg 10.5517/cc798yq) by combining numerous such observations, a way (and talent) that has largely lapsed these days.
A contemporary tackle this selective alkylation is perhaps to compute e.g. the wavefunction (ωB97XD/Def2-TZVPP/SCRF=water) of luteolin to examine the energies of the orbitals related to alkylation of the hydroxyl group, utilizing the power of the nucleophilic lone pair oxygen orbital (FAIR DOI: 10.14469/hpc/12185) as an indicator. The least secure such orbital (highest power) is often an indicator of essentially the most nucleophilic electron pair. On this case, the very best (most reactive) such orbital is the one adjoining to the carbonyl group, which thereby reveals a thriller, since it’s this very hydroxyl that resists alkylation! A transition state strategy to this is perhaps wanted to resolve the thriller, factoring in maybe steric results and so on.
| -0.6951 au | -0.7132 au |
|---|---|
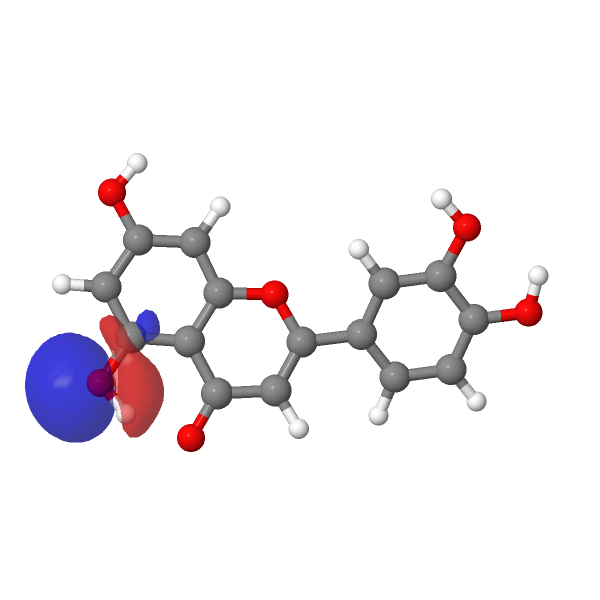 |
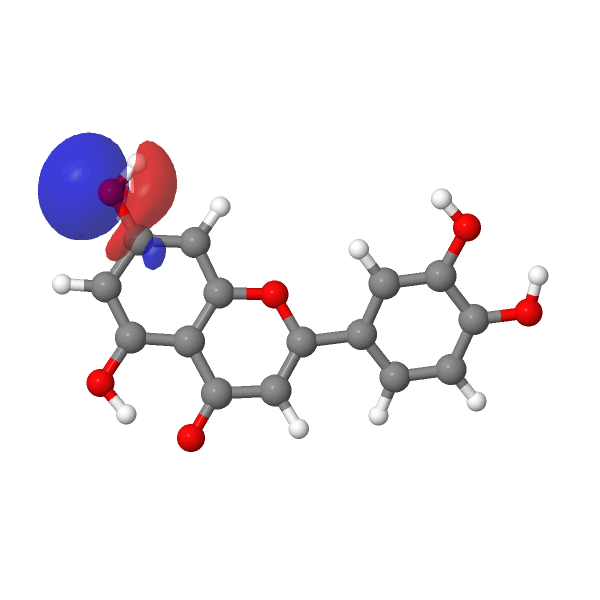 |
| -0.7169 au | -0.7205 au |
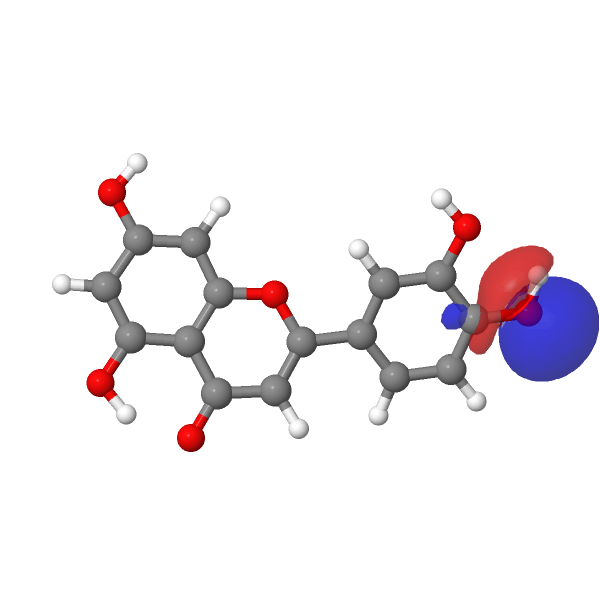 |
<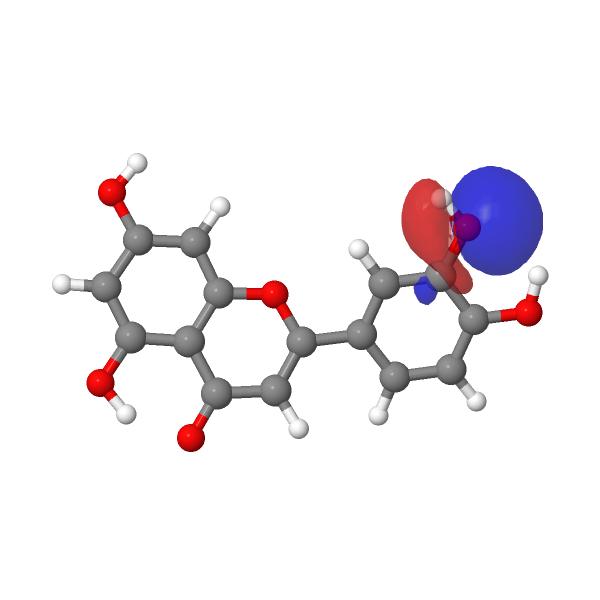 |
The calculated UV-Vis spectrum is proven under, displaying the height at ~300 NM answerable for the extreme yellow color (300-400 nm).
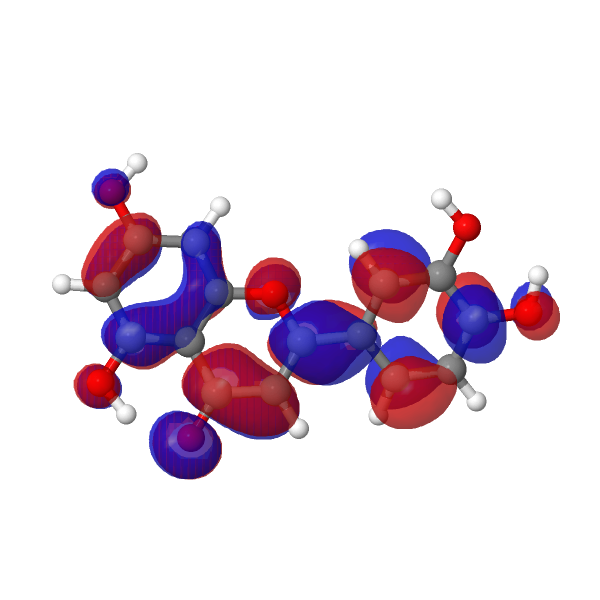
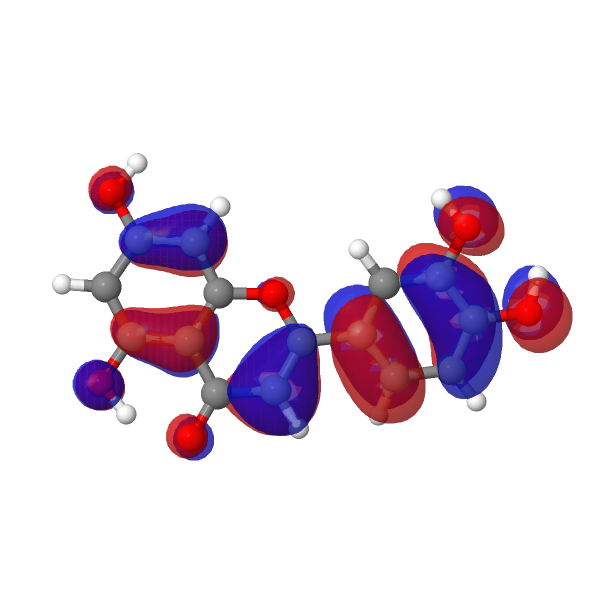
The strongest oscillator contribution to the transition is proven under.
| LUMO au | HOMO |
|---|---|
 |
 |
So right here I’ve solid just a little extra mild on this comparatively unknown pure yellow dye, that was used for a lot of centuries to color woollen supplies.
[ad_2]